PLOS Neglected Tropical Diseases: The Art of Writing and Implementing Standard Operating Procedures (SOPs) for Laboratories in Low-Resource Settings: Review of Guidelines and Best Practices

Inhaled isoflurane via the anaesthetic conserving device versus propofol for sedation of invasively ventilated patients in intensive care units in Germany and Slovenia: an open-label, phase 3, randomised controlled, non-inferiority trial -

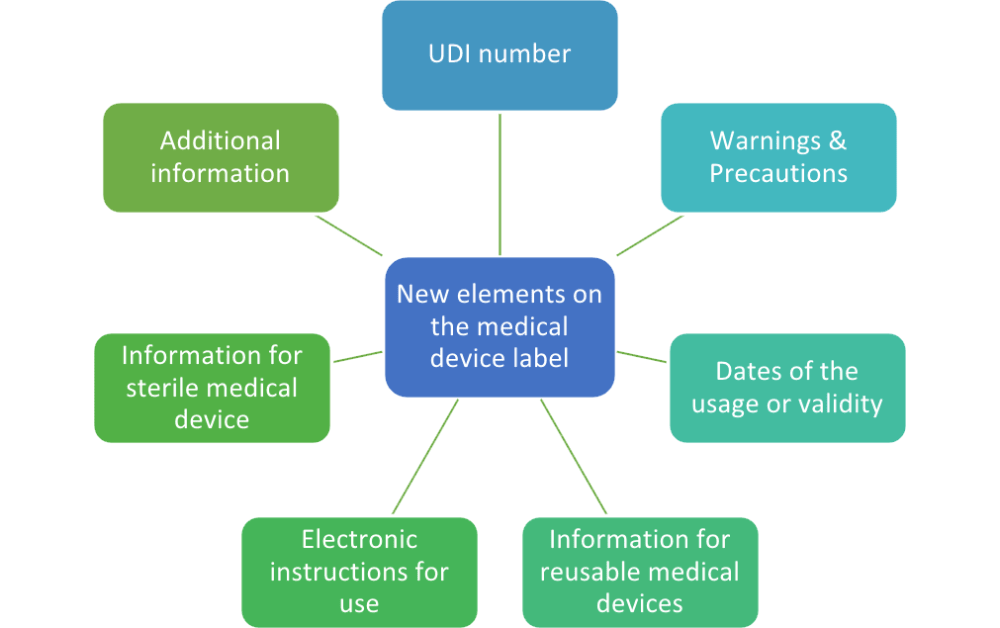
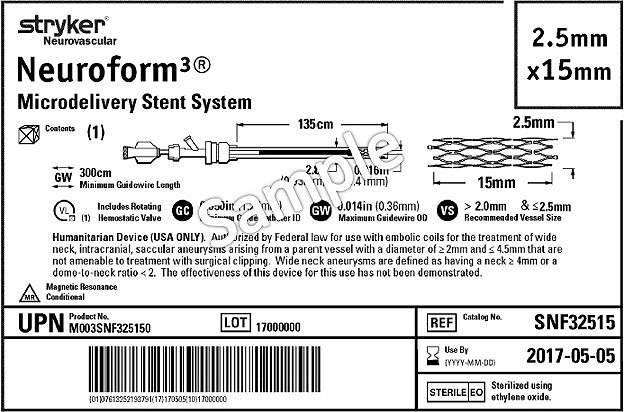
Guidance on Label and Instructions for Use for Medical Devices - FDA Regulatory Consulting and Training Services